 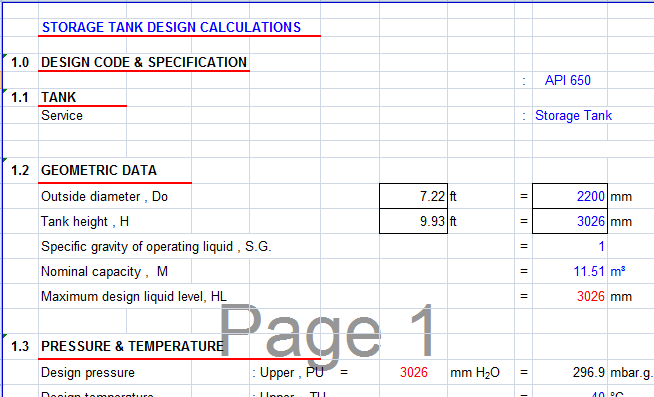 
Often the given constant is a combination of a number of meaningful physical quantities. But better would be to go back to the derivations of the equations and determining the proper units from there. You can do that simply by adding units that cancel the input units and add the output units. To get correct equations these missing units must be restored. The problem is that many of the constants in the formulae have, in fact, units, units which are unstated but implied by the units for the variables. 
I quite understand that including units in a calculation that was done without units is not a trivial task. If including units causes the answers to change, then either the inclusion of units was done incorrectly (the empirical equations were not properly adjusted) or the answers calculated without units were wrong. 697m (the unit is wrong rather than the value) it is still adding just a small amount. 697mm it is not just small, it is negligible - way smaller than your tolerances, or probably your measurement errors. The only difference is the size of the numbers.Ĭonsidering that you are working with lengths on the order of meters (about 20, overall) if L cri is indeed. Both large and small lengths can be expressed in either meters or millimeters. You cannot (meaningfully) add a number of meters to a number of millimeters. It makes no difference whether it increases the design liquid by a large amount or a small amount. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
